Research
Organic semiconducting materials for solar energy conversion
Contemporary organic photovoltaics uses emerging photoactive organic semiconducting materials for conversion of solar light into electricity. These materials are attractive because of low cost, unsurpassed mechanical flexibility and environmental innocence. There are several types of photovoltaic cells based on either organic semiconductors or their combinations with those inorganic, including dye-sensitized, bulk heterojunction, and supramolecular. Operation principles of these cells are very much alike. That is, a simple organic solar cell contains two organic semiconductor electrodes, one serving as an electron donor and the other as an electron acceptor. There are a few fundamental steps of photocurrent generation mechanism, common for all solar cell types (Scheme 1).
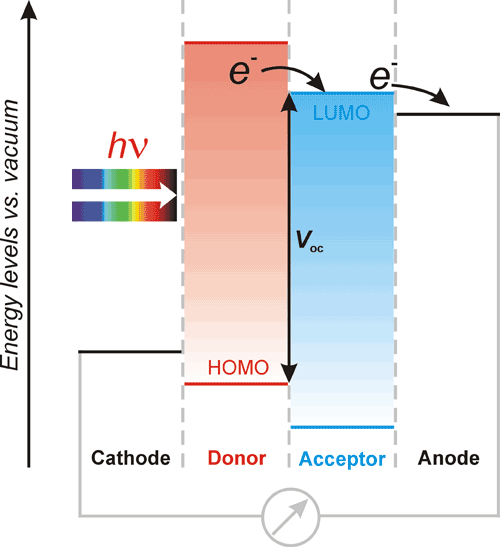
Scheme 1. A simplified energy diagram and operation principle of an organic solar cell. Voc stands for open circuit voltage.
In the simplest case, three steps are required for conversion of solar light into electricity including (i) absorption of light by a photoactive electron donor material at the cathode, (ii) photoinduced charge separation in this material leading to generation of charge carries, and (iii) collection of electrons by an electron-acceptor material at the photoanode with simultaneous injection of electrons from photocathode to reduce the photooxidized electron-donor material.
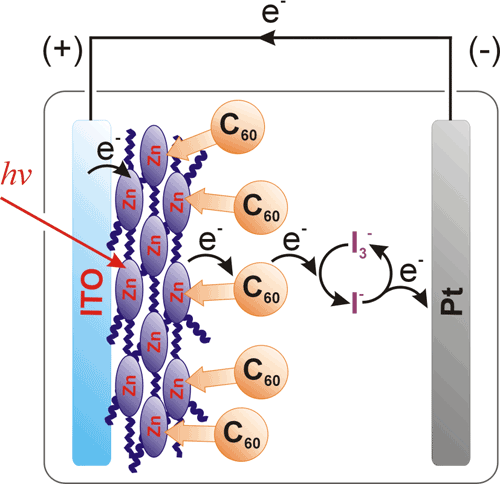
Scheme 2. A research solar cell assembly. In this case, the open circuit potential value and current direction depend upon the relative electron energy level of the redox mediator; the mediating redox couple here is I3-/3I-. (Reprinted with permission from Subbaiyan, N. K.; Obraztsov, I.; Wijesinghe, C. A.; Tran, K.; Kutner, W.; D’Souza, F. Supramolecular Donor−Acceptor Hybrid of Electropolymerized Zinc Porphyrin with Axially Coordinated Fullerene: Formation, Characterization, and Photoelectrochemical Properties J. Phys. Chem. C, 113, 8982–8989. Copyright 2009 American Chemical Society.)
For the research purpose, a simplified construction of an organic solar cell may involve an electrolyte solution and a redox charge mediator (Scheme 2). Charge mediation compensates low conductivity of organic semiconductors. This construction allows for fast evaluation and optimization of a solar cell design (Scheme 3).
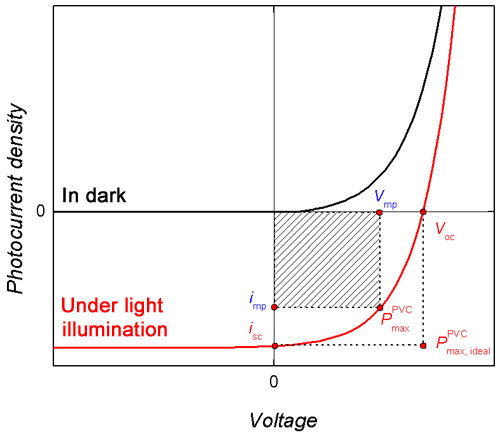
Scheme 3. Illustrative photocurrent-voltage curves for a solar cell in the dark and under light illumination. Voc – open circuit voltage, Vmp and jmp – voltage and photocurrent, respectively, corresponding to maximum power of the solar cell, jsc – short-circuit current, P PVCmax – maximum power of the solar cell, P PVCmax, ideal – maximum power of an “ideal” solar cell, i.e., that which converts all photogenerated charge to energy and transfers it into an external circuit.1
Our group investigates photoactive semiconducting materials based on the MeN4 complexes electron donors and [C60]fullerene derivative acceptors.2–4
References
-
Brabec, C.; Scherf, U.; Dyakonov, V. Wiley VCH: Weinheim 2008, pp 597.
-
Noworyta, K.; Marczak, R.; Tylenda, R.; Sobczak, J. W.; Chitta, R.; Kutner, W.; D’Souza, F. Langmuir 2007, 23, 2555–2568.
-
Marczak, R.; Sgobba, V.; Kutner, W.; Gadde, S.; D’Souza, F.; Guldi, D. M. Langmuir 2007, 23, 1917–1923.
-
Obraztsov, I.; Noworyta, K.; Hart, A.; Gobeze, H. B.; Kc, C. B.; Kutner, W.; D’Souza, F. ACS Appl. Mater. Interfaces 2014, 6, 8688–8701.



